
Europe’s Promising (But Rocky) Path to Embracing Single-Use Device Reprocessing
A proposed amendment to existing legislation could create centralized and harmonized rules for single-use device reprocessing across all EU member countries.

A proposed amendment to existing legislation could create centralized and harmonized rules for single-use device reprocessing across all EU member countries.
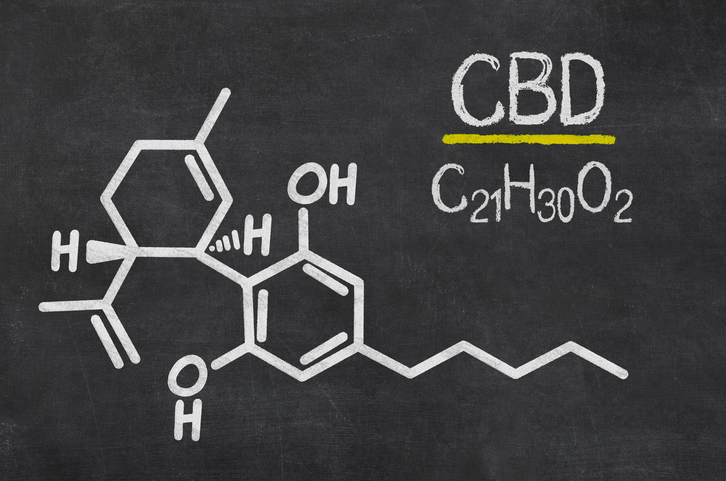
There must be clear rules and regulations in effect if we are to see medical-grade CBD become widely accepted amongst clinicians.

In today's dynamic environment, companies are navigating deal-making and innovation spurred by the vertical integration of AI and a rebounding IPO market. Here are ten global trends poised to define the industry in the coming year.

A handful of states have issued guidance documents that illustrate how regulators are viewing the IV hydration and IV therapy businesses, which infusion providers should consider as part of their overall compliance monitoring efforts.
We should never wait for tragedies to force a conversation around clear governance and accountability. Oversight has to evolve alongside innovation to protect people before harm occurs.

Healthcare and life sciences are about to face unprecedented AI-driven regulatory changes that will reshape everything from research and development to drug approval submissions. Here are 10 steps healthcare and life sciences organizations should take to strategically prepare.

Small practices play a critical role in healthcare delivery, but they cannot continue to absorb ever-increasing administrative demands without consequences.

As manufacturers play an active role in the patients’ healthcare journey, the boundaries between manufacturers and healthcare providers/payers are becoming increasingly blurry.

If the guidelines for data sharing are not adhered to by providers, then all the work that has gone into revising this regulation will have been for nothing. But the road to compliance doesn’t have to be overwhelming. Here are three essential steps for healthcare leaders in navigating the soon-to-be Part 2 landscape.

It is imperative to understand the various regulatory variations globally, develop a strategy for new technologies, engage in harmonization activities, and implement risk management measures.

Technology often plays a critical role in strategies to combat rising costs while maintaining quality care. Here are four trends expected to accelerate in 2025.

This not only enhances the quality of care and patient satisfaction but also optimizes operational efficiency.

Three tips to succeed in a highly regulated environment

New York Governor Kathy Hochul released a proposed set of cybersecurity regulations that require hospitals to establish new policies and procedures to protect themselves from ever-intensifying cyber threats. The state also put aside $500 million in funding to help hospitals upgrade their technology systems to comply with these new rules.

Cost is a primary reason people avoid or delay care, even when they are insured. But increasingly consumers are talking with providers, shopping around and taking advantage of cost transparency tools to access the care they need.

Eleven health systems have joined the coalition, which aims to extend and expand the existing federal hospital-at-home waivers and create an advanced acute care-at-home delivery model at the Center for Medicare & Medicaid Innovation.